
What’s changed isn’t dentistry’s scope - it’s the context around it.
If you have ever Googled your symptoms at 11 p.m. and ended up down a sleep apnea rabbit hole, you have probably also discovered that the diagnostic options seem to range from "fill out a questionnaire" to "spend the night in a hospital with electrodes taped to your head."
The good news: there is now a wider and more accessible range of home screening tools than ever before - from decade-old symptom questionnaires to FDA-cleared smartwatches to a new generation of AI face scan tools that require nothing more than a selfie camera. The challenge is knowing what each tool actually does, how accurate it is, and critically which one is the right starting point for you.
This guide breaks down every major home sleep apnea screening method with honest accuracy data, real costs, and a clear side-by-side comparison so you can make an informed decision.
Bottom Line Up Front: No single at-home screening tool replaces a clinical diagnosis. But the right tool can tell you whether you need one and that step alone changes everything for the roughly 80% of people with OSA who have never been tested.
Why Home Sleep Apnea Screening Matters
Obstructive sleep apnea affects an estimated one billion people worldwide, according to a widely-cited systematic review in The Lancet Respiratory Medicine. In the United States alone, the American Academy of Sleep Medicine estimates nearly 30 million adults are affected yet the vast majority remain undiagnosed. The commonly cited figure is that around 80% of moderate-to-severe OSA cases go undetected.
The consequences of going undiagnosed are serious. Untreated OSA is independently associated with hypertension, cardiovascular disease, type 2 diabetes, stroke, cognitive impairment, and significantly elevated risk of motor vehicle accidents. Early identification and treatment with CPAP, oral appliances, or lifestyle changes can reverse or mitigate many of these risks.
The bottleneck is access. Polysomnography, the clinical gold standard requires a specialist referral, an overnight stay in a sleep lab, and costs between $1,000 and $3,500. Even home sleep apnea tests require a physician's prescription. For most people, the journey to diagnosis never starts because the first step feels too high.
That is where home screening tools come in, not to replace clinical diagnosis, but to lower the barrier enough that people who need testing actually seek it.
Questionnaires are the oldest and most widely used screening tools for OSA in clinical settings. The three most common are:
Accuracy
The STOP-BANG is the most widely validated questionnaire and performs best in clinical populations with suspected OSA. Across multiple independent studies and meta-analyses:
The ESS numbers are particularly telling: the scale identifies daytime sleepiness well (high specificity) but misses a large proportion of OSA cases (very low sensitivity). This is because many people with significant OSA do not recognise how sleepy they actually are rather they have adapted to chronically poor sleep quality over years or decades.
Key limitations
Cost and accessibility
Free. All three questionnaires are publicly available online and take 3–5 minutes to complete. They are widely used in primary care as a first triage step before referring for diagnostic testing.
Best for: People who want a very quick, zero-barrier first check especially in clinical settings. Weakest for non-obese individuals, women, and anyone whose OSA is driven primarily by anatomy rather than BMI or lifestyle factors.
Consumer wearables primarily smartwatches and smart rings have evolved significantly as sleep health monitoring tools. Unlike questionnaires, they collect objective physiological data overnight. The most commonly used devices for sleep apnea risk monitoring include:
Accuracy
Wearable accuracy varies significantly by what is being measured. For general sleep staging, a 2024 Brigham and Women's Hospital study published in Sensors found:
For sleep apnea detection specifically, a 2024 study found that newer-generation smartwatches correctly flagged OSA risk in 75–96% of users who were later confirmed to have OSA. However, performance drops significantly for mild OSA - wearables detect events better as severity increases.
An important caveat: wearables detect physiological consequences of OSA - breathing disruptions, oxygen desaturations, heart rate anomalies rather than the structural causes of OSA. This means they require the condition to already be causing nighttime events in order to detect it.
Key limitations
Cost and accessibility
Apple Watch: $399–$799. Oura Ring: $299 + $5.99/month subscription. Samsung Galaxy Watch: $299–$649. Withings Sleep Analyzer: $129. No prescription required for consumer versions; FDA-cleared diagnostic wearables (WatchPAT, etc.) require clinical prescription.
Best for: People who already own a compatible smartwatch, or who are willing to invest in overnight monitoring for ongoing tracking. Particularly useful for identifying active breathing disturbances and trending sleep quality over time. Less useful for identifying structural risk before symptoms develop.
AI face scan tools for sleep apnea screening such as FaceX by Soliish analyzes the structural features of your face using a convolutional neural network (CNN) trained on clinical datasets with polysomnography-confirmed OSA diagnoses. The scan takes a front-facing photograph through your phone or webcam and detects craniofacial risk markers associated with obstructive sleep apnea.
The science behind this approach is well-established: OSA is fundamentally a structural disorder. Anatomical features including retrognathia (a recessed jaw), neck dimensions, midface development, and palate shape determine upper airway geometry and upper airway geometry determines OSA risk. These features are measurable in standard photographs.
Accuracy
The AI selfie scan evidence base has grown rapidly, with multiple peer-reviewed studies now published:
What makes AI selfie scanning uniquely valuable
The key differentiator for AI face scanning is not just accuracy. It is what it measures:
The table below summarises the key attributes of each screening method. ✓ = strong capability ◑ = partial / conditional ✗ = limited or not applicable
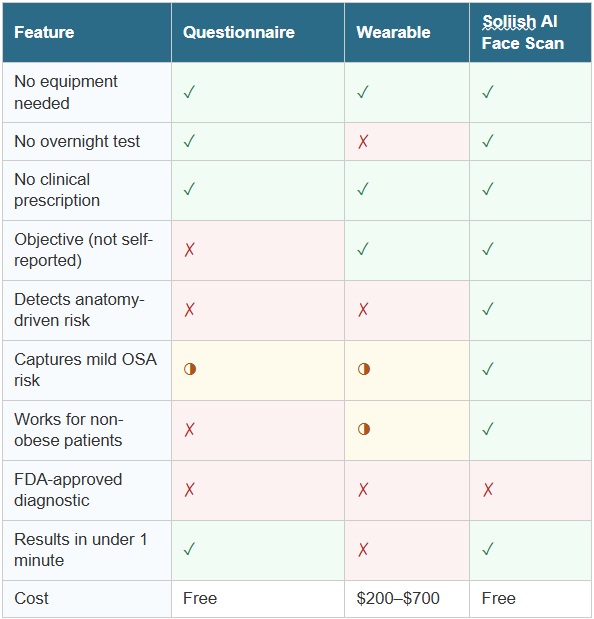
The honest answer is that most people will benefit from more than one tool at different stages of the screening-to-diagnosis journey. Here is a practical guide:
Start with an AI face scan if…
Add a questionnaire if…
Use a wearable if…
Recommended Pathway: AI face scan → discuss result with GP or dentist → HSAT if indicated → formal diagnosis → treatment. This pathway removes the gatekeeping barrier of the first step while ensuring every subsequent step is clinically grounded.
Every home sleep apnea screening method reviewed here has a legitimate role and honest limitations. Questionnaires are fast and free but subjective. Wearables provide objective overnight data but require the condition to already be causing detectable events. Home sleep tests are the closest thing to diagnostic gold standard at home, but require clinical involvement and are not screening tools.
AI face scanning fills a unique and underserved gap: it provides objective, structural risk assessment instantly, without equipment, without an overnight test, and without a prescription. It identifies the patients most likely to be missed by every other first-line screening method: those whose OSA is driven by anatomy, not lifestyle.
The future of sleep apnea screening is not a single tool, it is a layered, accessible pathway that starts where the patient actually is, and moves them efficiently toward the care they need. An AI face scan is the best place to start that journey.
1. Gao EY, et al. (2024). Artificial intelligence facial recognition of obstructive sleep apnea: a Bayesian meta-analysis. Sleep and Breathing, 29(1):36. doi: 10.1007/s11325-024-03173-3
3. Park JY, et al. (2025). Machine learning model for screening OSA risk using craniofacial photography with questionnaires. Journal of Clinical Sleep Medicine, 21(5):843-854. doi: 10.5664/jcsm.11560
4. Giorgi L, et al. (2025). Advancements in Obstructive Sleep Apnea Diagnosis Through Artificial Intelligence: A Systematic Review. Healthcare, 13(2):181. doi: 10.3390/healthcare13020181
5. Chiu HY, et al. (2017). Diagnostic accuracy of the Berlin questionnaire, STOP-BANG, STOP, and Epworth sleepiness scale: A bivariate meta-analysis. Sleep Medicine Reviews, 36:57-70. doi: 10.1016/j.smrv.2016.10.004
7. Robbins R, et al. (2024). Accuracy of three commercial wearable devices for sleep tracking in healthy adults. Sensors, 24(20):6532. doi: 10.3390/s24206532